Gaining Headway: GW Pharmaceuticals Carves a Niche for Cannabinoid Medicines

By Oliver Bennett, Special Contributor, New Frontier Data
The progress of medical cannabis in Europe has been marked by small steps rather than big leaps. Currently, the consumption of cannabis for medical is legal in 22 European countries, though there are regional and national differences and a variety of systems across the European Union (EU) characterized by constraints and inconsistencies. Meanwhile, recreational cannabis remains illegal in most European states, while being legal in the Netherlands and decriminalized in others like Germany, Portugal, the Czech Republic, and Spain.
The outlook for Europe’s medical cannabis programs is also somewhat haphazard. Some countries such as Denmark and France have established national medicinal cannabis pilots, while six others – Austria, Belgium, France, Slovenia, Spain, and Romania – have only legalized selected cannabis-based pharmaceuticals. Nevertheless, while they are in some respects more cautious than the U.S. and Canada, many European governments are open to research – with growth showing no sign of abating.
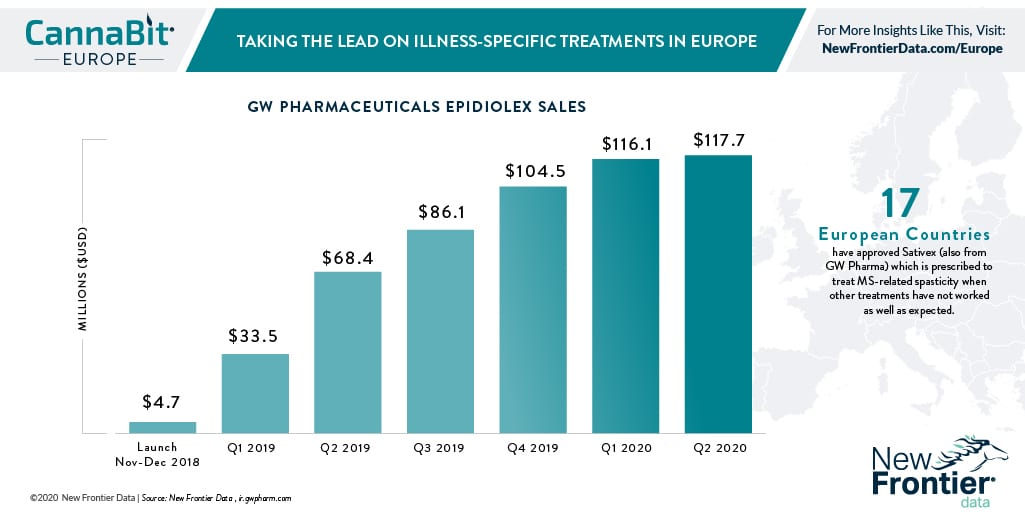
In Europe, medical cannabis is increasingly recognised for a panoply of ailments including neurological diseases, chemotherapy-related nausea, inflammatory diseases, pain, and epilepsy, to mental health conditions such as anxiety and depression. The pharmaceutical brand with the biggest traction is Epidiolex from U.K.-based GW Pharmaceuticals, a success story across Europe as a popular prescription drug for patients with Lennox-Gastaut syndrome or Dravet syndrome, two forms of epilepsy.
Sativex (also known as nabiximols, also from GW Pharma), has been approved in 17 European countries, and is now available in nine of them. It is also prescribed to treat MS-related spasticity, if typically only after other attempted treatments have been exhausted (it must be used in tandem with another anti-spasticity medication). The artificial cannabinoid Nabilone is used for the nausea associated with chemotherapy – again, only after other treatments have proven unhelpful or unsuitable. Such examples suggest that there is market potential in designing products with specific illnesses in mind.
Sometimes the gap between medical and recreational cannabis usage seems inconsistent. Italy, for example, has decriminalized recreational cannabis while maintaining strict protocols towards medical cannabis. Yet, many Italians already use medical cannabis, after what one newspaper deemed Italy’s cannabis mania boomed following a December 2016 edict allowing hemp production. The annual consumption of legal medical cannabis grew from 40 kilograms in 2013, to nearly 10x more produced by Florence’s Military Chemical Pharmaceutical Plant during its 2017 start. To date, eligibility for medical cannabis in Italy is limited to MS, chronic pain, glaucoma, Tourette’s syndrome, nausea, spinal cord injury, anorexia, and cachexia – along with treatment for HIV/AIDS, chemotherapy, or radiation treatments (again, only after previous treatments had otherwise failed).
Germany has more than 60,000 people registered to receive medical cannabis. Demand has soared since the 2017 decision allowing doctors to prescribe medical cannabis flowers and cannabis extract. German patients must get authorisation from a medical doctor, though there are no qualifying conditions required, and the country’s Federal Institute for Drugs and Medical Devices (BfArM) has a cannabis-specific agency overseeing the cultivation and production of medical cannabis. BfArM allows doctors to use their discretion, and the key ailments prescribed with cannabis are pain, spasticity, anorexia, epilepsy, ADHD and Tourette’s syndrome. The German approach is approved of by the new European Medicinal Cannabis Association (EUMCA).
France last year announced plans to commence a medicinal cannabis pilot (if currently delayed), to run for two years with a sample of 3,000 patients. Prescriptions for medicinal cannabis are expected to begin in January 2021 but on two conditions: that patients have exhausted all other options, and treatment falls into an approved list including child epilepsy, MS, palliative care, chemotherapy in late-stage cancer, and chronic pain. A government commission on cannabis regulations includes a working group of 33 French members of parliament, while the Agence Nationale de Sécurité du Médicament, has been praised for stewarding “the first French data on the efficiency and safety” of medical cannabis. The lead agency L630 advises the government alongside ACT Collective, an organization of French patient groups. Once the two-year pilot is concluded, the French government will decide whether to extend or expand it.
Since November 2018, when the U.K. government allowed the legal prescription of cannabis-based medicinal products (CBMPs), there have only been around 400 private prescriptions and two NHS prescriptions for medical cannabis. Recreational cannabis remains illegal, and only specialist registered doctors are allowed to prescribe cannabis (and only then for selected issues including epilepsy, chemotherapy nausea and multiple sclerosis, as per NHS guidelines).
Yet, a survey suggesting that 1.4 million Britons are using illicit cannabis to self-medicate believes that the highest numbers of those include people either with Huntington’s disease, schizophrenia, or Parkinson’s disease, or else depression, anxiety, and/or chronic pain – with the most common mental-health complaint being depression.
Epidiolex this year has been available for British epilepsy sufferers since January, and Sativex is approved to treat MS patients. Along with a £1.2million UK study assessing the effect of CBD on Parkinson’s disease, 20,000 U.K. patients are to be given medical cannabis over a two-year period in an initiative to gather Europe’s largest such study of the drug. The independent study led by Drug Science during Project Twenty21 will see patients supplied with subsidised cannabis products by the end of 2021, and study the effects on patients with chronic pain, epilepsy, multiple sclerosis, post-traumatic stress disorder (PTSD), Tourette’s syndrome, anxiety disorder, or those with a history of substance misuse.
Spain’s program is also making steps forward. Proponents in Catalonia are lobbying the Spanish government to legalize medical cannabis, while also aiming to end informal “cannabis clubs”, i.e., non-profit cooperatives for self-medicating patients.
There is a growing interest in the European medical cannabis business, and established companies are hovering. Canopy Growth has been given permission to import medical cannabis to its distribution centre in the U.K. Aurora last year signed an agreement to supply Italy with medical cannabis, and Tilray hopes to make its Portuguese facility in Cantanhede a base to set up a European supply chain. In Denmark, some cultivators have already started growing cannabis after a change in regulations was designed to diversify the supply chain since demand outstrips supply in larger European markets, and the EU relies on imports from Canada.
There remains a regulatory dance across key European countries with a poor supply chain, high prices and legal inconsistencies. As well as the need for scientific evidence to inform regulation and prescription and gain the EU’s good manufacturing practice (GMP) certification, the legalization of medical cannabis is seen as a harbinger for the legalization of recreational use, which concerns policymakers.
And while there are strict European controls on how cannabis-based drugs are manufactured, there is a lack of harmonised quality standards. It is argued that a strong EU regulatory framework on medical cannabis is needed so that investors have stability – and patients some consolation in clarity for their expectations and treatment.


