DEA’s Proposed Interim Rules Throw CBD Industry into Legal Jeopardy
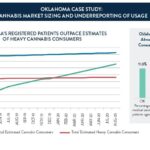
Oklahoma Case Study: Cannabis Market Sizing and Underreporting of Cannabis Usage (Cannabit Graphic)
August 24, 2020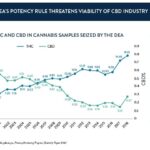
DEA’s Potency Rule Threatens Viability of CBD Industry
August 30, 2020By Trevor Yahn-Grode, Data Analyst, New Frontier Data
On August 20, the Drug Enforcement Agency (DEA) published its much-anticipated Interim Final Rules (IFR) regarding hemp production, regulations designed to put the agency in compliance with the 2018 Farm Bill. Included in the IFR is a revision of the agency’s definition of THC and marijuana extracts (excluding hemp products containing less than 0.3% THC), along with the removal of FDA-approved, CBD-based medications from a schedule V listing.
Significant controversy has arisen, however, over the DEA’s interpretation of THC content as it relates to hemp cannabinoid processing. The agency stated:
“The definition of hemp does not automatically exempt any product derived from a hemp plant, regardless of the Δ,9 -THC content of the derivative. In order to meet the definition of ‘hemp,’ and thus qualify for the exemption from schedule I, the derivative must not exceed the 0.3% Δ,9-THC limit.”
While seemingly innocuous, the rule has drawn widespread criticism from CBD processors, who point out that it effectively criminalizes key parts of the CBD production process.
To synthesize CBD or other cannabinoids from hemp, hemp biomass must first go through an extraction process. While various extraction methods exist, all of them result in a temporary increase in the concentration of Delta-9 THC, and it is almost impossible to ensure that a given concentration will not exceed the requisite 0.3% limit. Although the work-in-progress extract is eventually diluted and refined to contain less than 0.3% THC before being put into consumer products, the DEA’s rules mean that processors would be in possession of a schedule I controlled substance during much of their extraction process.
That goes beyond saying that end-use products must contain less than 0.3% THC, but rather makes the process of creating such products de facto illegal. If enforced, the rules would make it almost impossible for a company to manufacture CBD for consumer use without holding a research license from the DEA.
Though the policies laid out by the interim rules are not final (the DEA will be taking comments from industry stakeholders through October 20), the DEA insists that they have been in effect since hemp was federally legalized in 2018, meaning both that all major CBD processors are currently in violation of the DEA’s rules, and in possession of schedule I controlled substances. While it is far too early to fully understand the impact which the rules would have on the industry, it is clear that – should they be enforced as currently written – hemp processors could face real criminal risk under the Controlled Substances Act.